The global demand for minimally invasive surgery (MIS) has positioned China as a pivotal hub for medical technology manufacturing. As a leading exporter, we recognize that the "China Top Endoscope Factory" designation requires more than just production capacity; it demands a relentless focus on optical precision, supply chain resilience, and rigorous regulatory compliance.
China's Factory 4.0 initiatives have revolutionized the medical device sector. By integrating IoT-driven quality management systems and automated assembly lines, manufacturers can now ensure the structural integrity and reliability of endoscopes and peripheral equipment. This digital transformation guarantees that every unit—from high-frequency electrosurgical units to precision suction pumps—meets international ISO 13485 and FDA standards.
Healthcare providers globally are moving away from monolithic procurement strategies, opting instead for agile supply chains. Emfix Medical leverages its mature production infrastructure to offer:
Headquartered in Shanghai, China, often referred to as the "Oriental Paris," Shanghai Emfix Medical Co., Ltd. has been at the forefront of the medical device industry for over 30 years. Our journey reflects the broader growth of China's high-tech manufacturing sector, blending historical expertise with modern innovation.
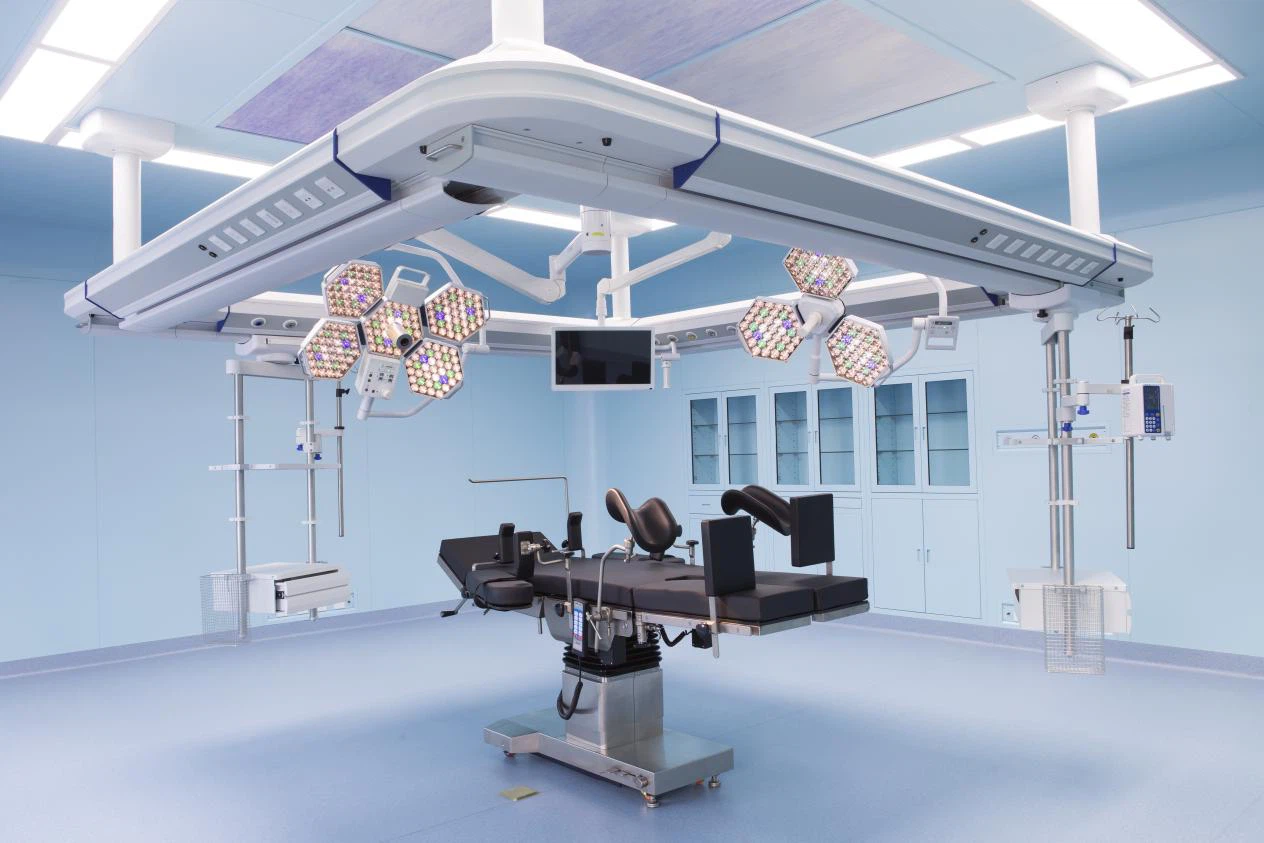
We manage three state-of-the-art factories. Unlike trading companies, we manage every phase from R&D to manufacturing, ensuring zero third-party interference.
With products in over 50 countries, we maintain strict adherence to international medical standards (CE, FDA, MDA). We are a recognized Class A Enterprise in China, reflecting our leadership position.
Our mission is to provide innovative medical solutions that improve patient outcomes. We focus on energy-efficient manufacturing processes to contribute to a sustainable global healthcare environment.
A: We utilize strict internal quality control protocols, including automated testing for our suction/irrigation pumps and electrosurgical units, ensuring compliance with ISO 13485 standards before any product leaves the factory.
A: Yes. With over 30 years of experience, we support OEM/ODM services, adapting power requirements, user interface languages, and regulatory certifications to suit your country's specific needs.
A: We have a dedicated logistics team experienced in shipping sensitive medical equipment worldwide, ensuring appropriate packaging to maintain equipment calibration during transit.